Elleth, I couldn't agree more and I'm certainly not suggesting anyone just uses a modern fire steel and calls it dwarven. The only reason I'm posting this now is because I'd already written almost all of it and saved it as a draft

----------------------------------------------------------------------------------------
Very difficult question, first up, in a fantasy story I wrote when I was 16 (Not set in middle earth) I had a race of people who used magic to make lightening storms and harnessed it to electrolyse and isolate certain metals. Perhaps the Blue wizards could do similar


Anyways, joking aside, I wondered if it is possible to isolate ether cerium or magnesium metal without electricity, so I dug out my chemistry textbooks and hit the internet:
Concerning cerium:
Cerium was discovered in 1803 by Jacob Berzelius and Wilhelm von Hisinger in Sweden, and independently in the same year by Martin Klaproth in Germany.
Berzelius and Hisinger discovered the new element in a rare reddish-brown mineral now known as cerite, a cerium-lanthanide silicate.
Berzelius and Hisinger prepared cerium salts from cerite and investigated their chemical reactions. Although they could not isolate the pure metal, they found that cerium had two oxidation states: one yielding colorless salts, the other yellow-red ones.
Carl G. Mosander, who worked closely with Berzelius, prepared metallic cerium in 1825. First he reacted cerium sulfide with chlorine, yielding anhydrous cerium chloride. He reduced the chloride with potassium, forming potassium chloride and metallic cerium. He burnished the resulting brown powder to obtain a gray material with a dull metallic luster – cerium, although rather impure.
https://www.chemicool.com/elements/cerium.html
So, although Mosander's cerium was impure, it would still have been pure enough for Ferrocerium.
Concerning Magnesium:
Magnesium was first isolated in 1755 by Joseph Black, using electrolysis, however, the following process is now used in industry to separate Magnesium metal from dolomite ore:
Thermal Reduction Process:
Dolomite is normally treated by the thermic method: this consists of reducing magnesium oxide, produced by calcining the dolomite raw material, with ferrosilicon to produce metallic magnesium and a calcium iron silicate slag.
Step 1: Dolomite ore is crushed and heated in a kiln to produce a mixture of magnesium and calcium oxides, a process known as calcining.
Step 2: The next step is reduction of the magnesium oxide. The reducing agent is ferrosilicon (an alloy of iron and silicon) which is made by heating sand with coke and scraps iron, and typically contains about 80% silicon.
Step 3: The oxides are mixed with crushed ferrosilicon, and made into briquettes for loading into the reactor. Alumina may also be added to reduce the melting point of the slag. The reaction is carried out at 1500 - 1800 K under very low pressure, close to vacuum. Under these conditions the magnesium is produced as a vapour which is condensed by cooling to about 1100 K in steel-lined condensers, and then removed and cast into ingots:
Step 4: The forward reaction is endothermic and the position of equilibrium is in favour of magnesium oxide. However, by removing the magnesium vapour as it is produced, the reaction goes to completion. The silica combines with calcium oxide to form the molten slag, calcium silicate.
The process gives magnesium with up to 99.99% purity, slightly higher than from the electrolytic processes.
http://metalpedia.asianmetal.com/metal/ ... tion.shtml
The only part of this that definitely can't be recreated in middle earth is the 'close to vacuum' conditions, however this is only used to reduce the temperature the reaction needs to be carried out under and reduce the energy needed, it isn't impossible at 1 atmosphere, it would just need to be hotter.
Conclusion:
Both metals are produced using electricity now for one simple reason, it's easier. That doesn't mean you can't get them without if you have the skills and equipment. The question we have to ask now is, what chemistry was available in Middle Earth?
Dwarves probably couldn't have mined as deep as they did without pyrotechnics, and since we don't know weather Tolkien intended mithril to be an existing element that we now have access to or something purely magical and unique to middle earth I won't go into that.
There are some clues in the text, the biggest one is Gandalf's fireworks:
The finest rockets ever seen:
they burst in stars of blue and green,
or after thunder golden showers
came falling like a rain of flowers.
For this I'll assume Gandalf's fireworks were created with a combination of chemistry and pyrotechnics, not magic. The main ingredient to fireworks is gunpowder (Potassium nitrate, sulfur and carbon) which is fairly easy to make, ''golden showers'' would be iron filings, also easy. However ''stars of blue and green'' isn't quite so simple. Blue is from copper (II) chloride, and green, barium (II) chloride.
Copper (II) Chloride is made in in industry now by reacting copper metal (available in middle earth) with chlorine gas. The only non-electric way to make chlorine gas is to oxidise hydrochloric acid, that reaction need's copper (II) chloride (which your trying to make and don't have yet) as a catalyst so isn't viable. There are lot's more ways to make the chemical in a lab, and only one that I think is viable in middle earth:
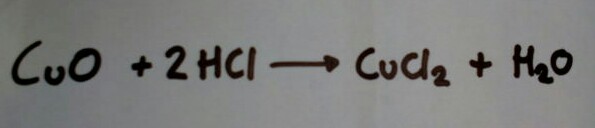
- IMG_20180910_182929.jpg (11.63 KiB) Viewed 15774 times
Where copper (II) oxide is dissolved in hydrochloric acid to make copper (II) chloride and water. The only problem with this is copper (II) oxide isn't a common copper ore, fortunately it does occur naturally as a mineral called tenorite which happens to be a volcanic sublimate and would, more than likely, be found in the lonely mountain (which can only be an extinct volcano).
Barium (II) chloride is slightly more complex, given that it's a two stage reaction although it can be made straight from normal barium ore:
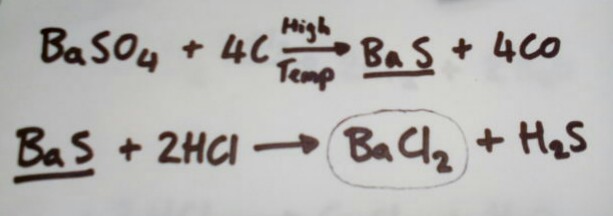
- IMG_20180910_182920.jpg (21.45 KiB) Viewed 15774 times
Where Barite ore is reacted with carbon at high temperature, then the product is dissolved in hydrochloric acid.
Both the above reactions are much more simple and have fewer steps than what would be needed to produce metallic magnesium or cerium. The immediate hitch with cerium is you need chlorine gas (which could, at a push, be produced using copper (II) chloride as a catalyst since that is available) so cerium is less likely than magnesium.
In my opinion the equipment/know how to make magnesium in a lab in middle earth would be there and cerium, maybe? However that is in a lab and in small amounts. Mass-production/availability to anyone other that the very rich and privileged is unlikely.
Hope this was useful and not to in depth

and If any of it's wrong, feel free to correct me, It's been a while
